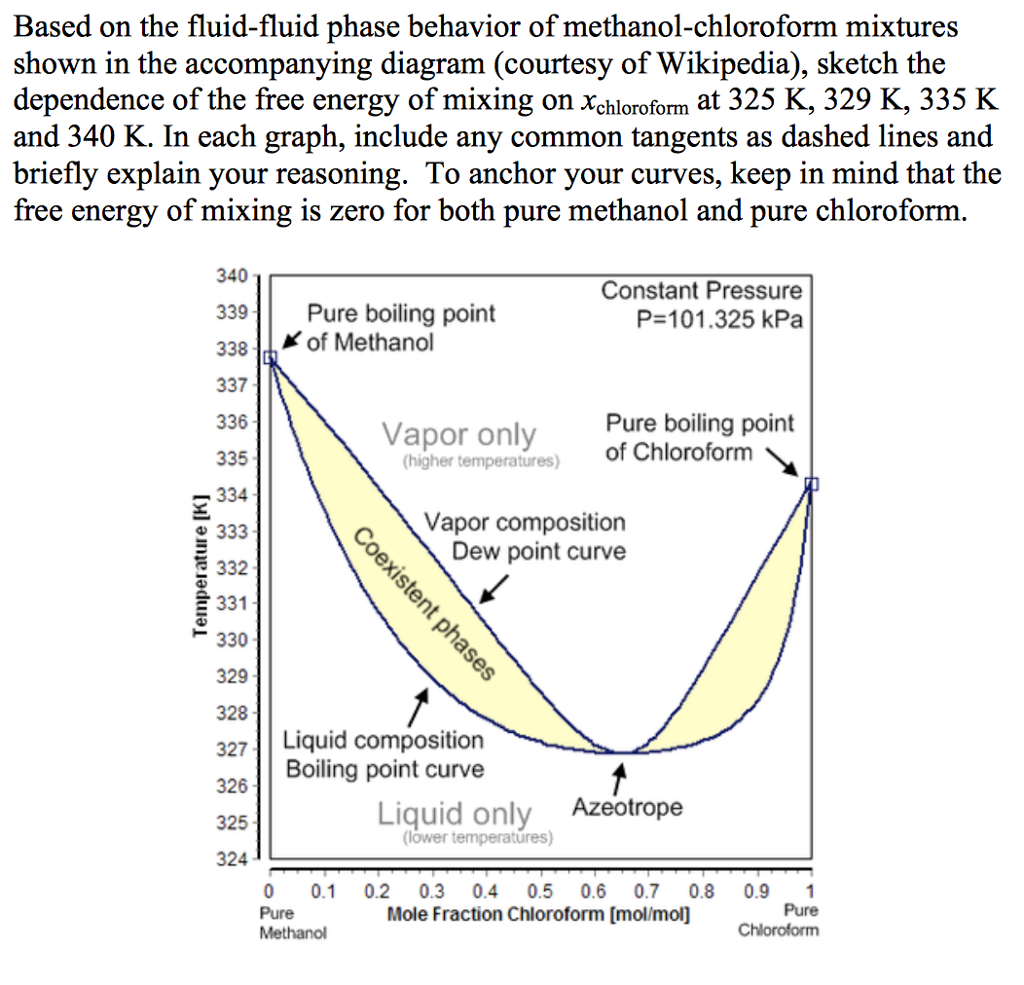
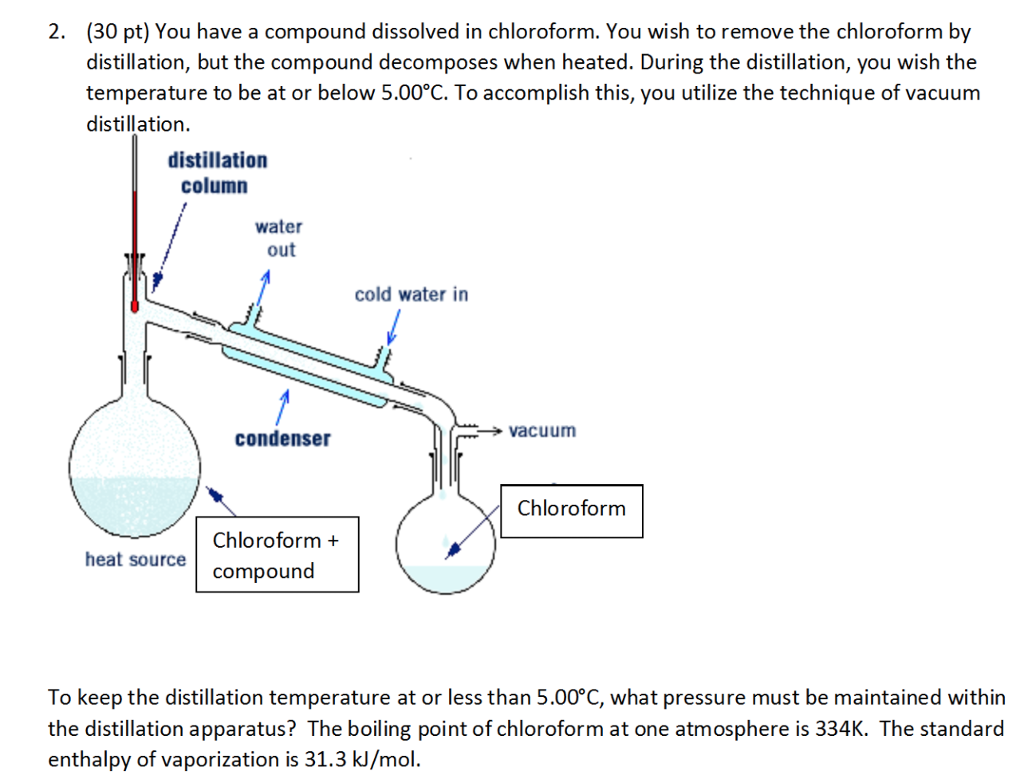
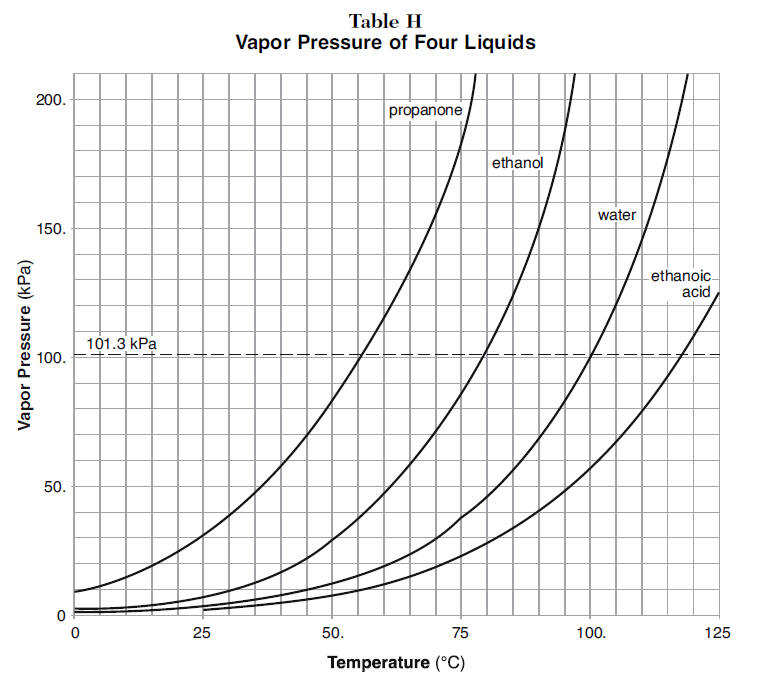
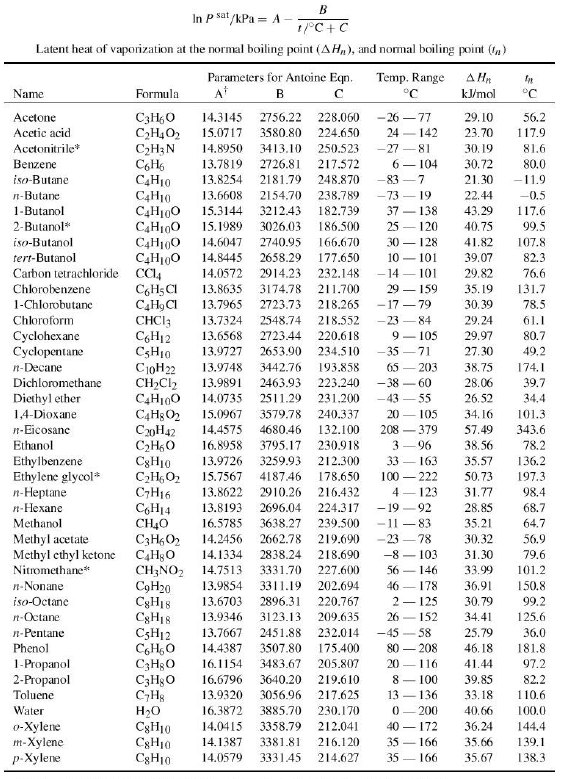
Separation of methanol-chloroform mixture using pressure-swing distillation: Modeling and optimization

Normalized UV–vis absorption spectra in a) chloroform solution and b)... | Download Scientific Diagram

Comparison of extractive distillation and pressure-swing distillation for acetone/chloroform separation - ScienceDirect
![Density, Viscosity, Vapor−Liquid Equilibrium, and Excess Molar Enthalpy of [ Chloroform + Methyl tert-Butyl Ether] | Journal of Chemical & Engineering Data Density, Viscosity, Vapor−Liquid Equilibrium, and Excess Molar Enthalpy of [ Chloroform + Methyl tert-Butyl Ether] | Journal of Chemical & Engineering Data](https://pubs.acs.org/cms/10.1021/je100821g/asset/images/medium/je-2010-00821g_0003.gif)
Density, Viscosity, Vapor−Liquid Equilibrium, and Excess Molar Enthalpy of [ Chloroform + Methyl tert-Butyl Ether] | Journal of Chemical & Engineering Data

Applied Sciences | Free Full-Text | Effect of Ionic Liquids on the Isobaric Vapor-Liquid Equilibrium Behavior of Acetone-Chloroform
What is the normal boiling point of chloroform if its heat of vaporization is 31.4 kJ/mol and it has a vapor pressure of 190.0 mmHg at 25.0 °C? - Quora