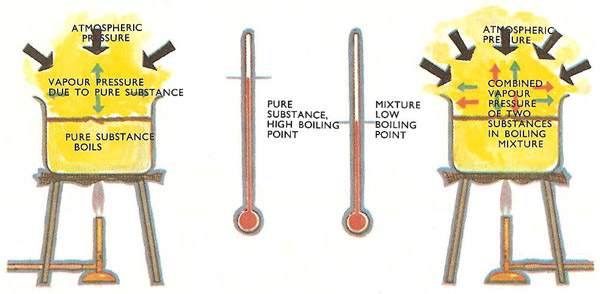
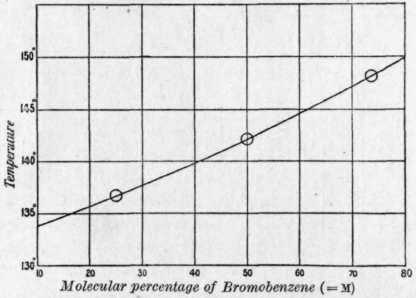
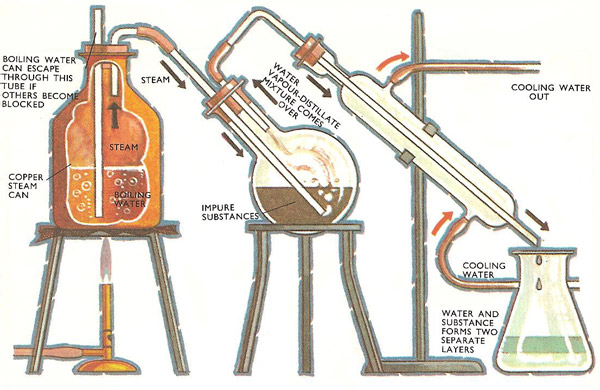
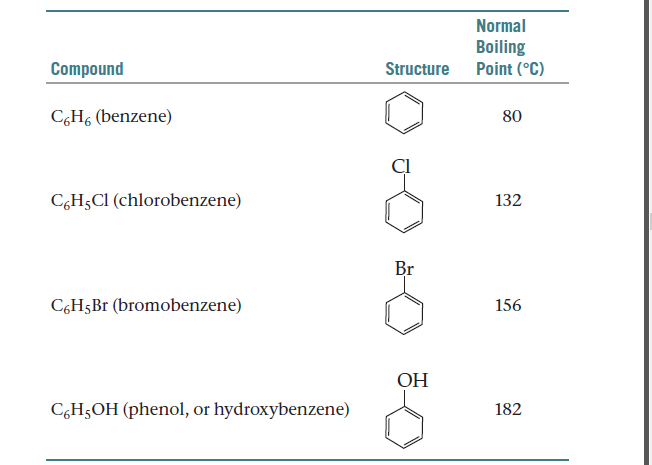
1. A mixture is chlorobenzene and water (which are virtually immiscible) boils at 90.3^° C at an external pressure of 740.2mm. The vapour pressure of pure water at 40.3^° C if 530.1mm.

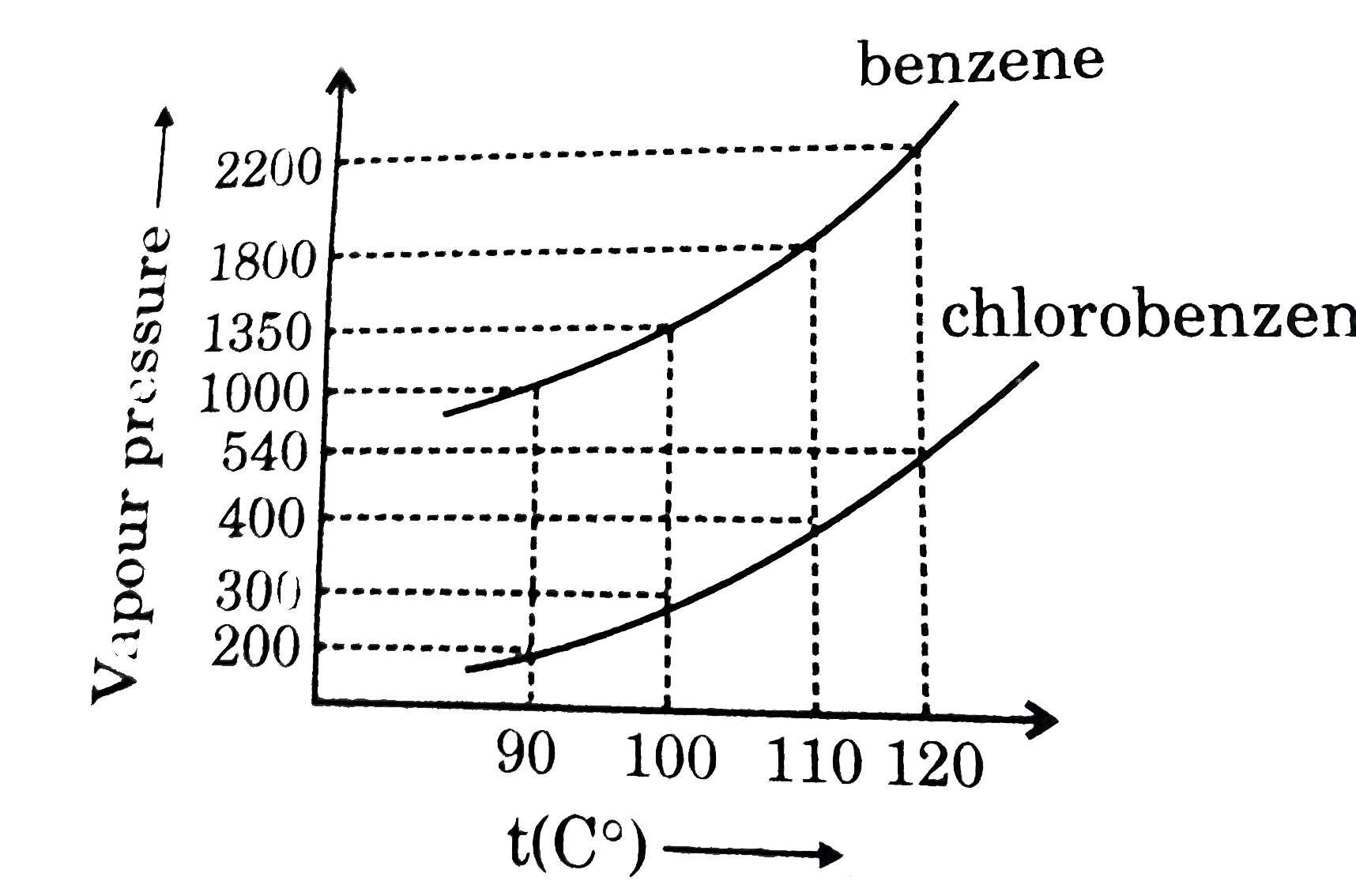
Assuming the formation of an ideal solution, the boiling point of a mixture containing 1560 g benzene (molar mass = 78) and 1125 g chlorobenzene (molar' mass = 112.5)' using the given
What is the order for the boiling point for ortho-chlorobenzene, meta- chlorobenzene and para-chlorobenzene? - Quora

Boiling point T b ,°С, vs. molecular weight M: (1): (a) benzene, (b)... | Download Scientific Diagram

Correct order of boiling point for the given compounds is:a) 1 - chloropropaneb) isopropyl chloridec) 1 - chlorobutane
What is the order for the boiling point for ortho-chlorobenzene, meta- chlorobenzene and para-chlorobenzene? - Quora

Q. Why boiling point of ortho dichloro benzene is greater then para & ortho dichloro benzene? - YouTube
Using the following information determine the boiling point of a mixture contains $1560$ gm benzene and $1125\; gm$ chlorobenzene, when the external pressure is $1000\; torr$ Assume the solution is ideal.Given :

SOLVED: ii A mixture of liquid chlorobenzene and water is boiling at 130 mm Hg pressure. Liquid chlorobenzene is immiscible in water.Determine the boiling point of the mixture in K and the
What is the order for the boiling point for ortho-chlorobenzene, meta- chlorobenzene and para-chlorobenzene? - Quora
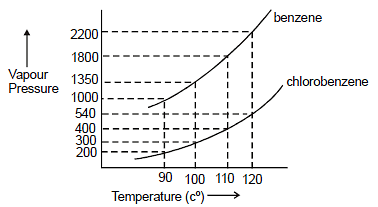
Assuming the formation of an ideal solution, determine the boiling point of a mixture containing 1560 g benzene (molar mass = 78) and 1125 g chlorobenzene (molar mass = 112.5) using the