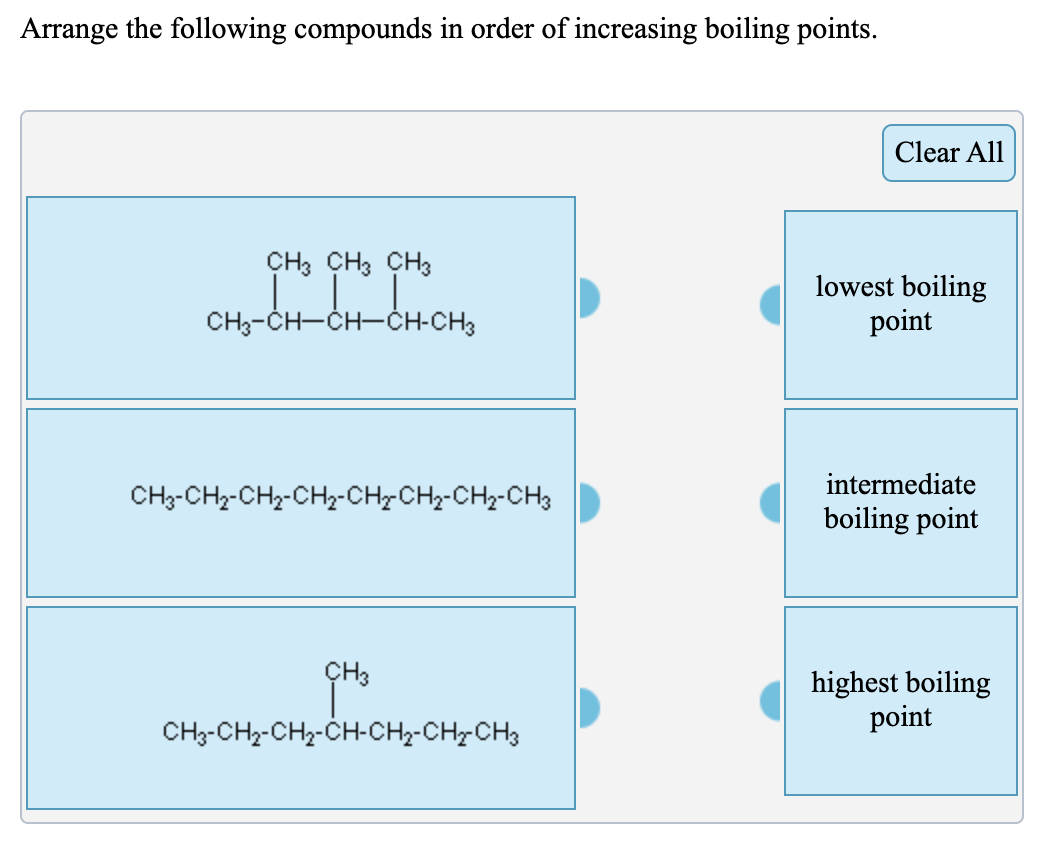
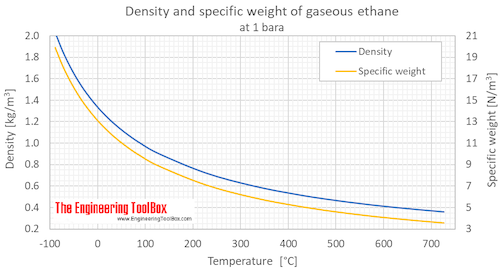
SOLVED:Pick the compound with the highest boiling point in each pair. Explain your reasoning. a. CH3OH or CH3SH b. CH3OCH3 or CH3CH2OH c. CH4 or CH3CH3
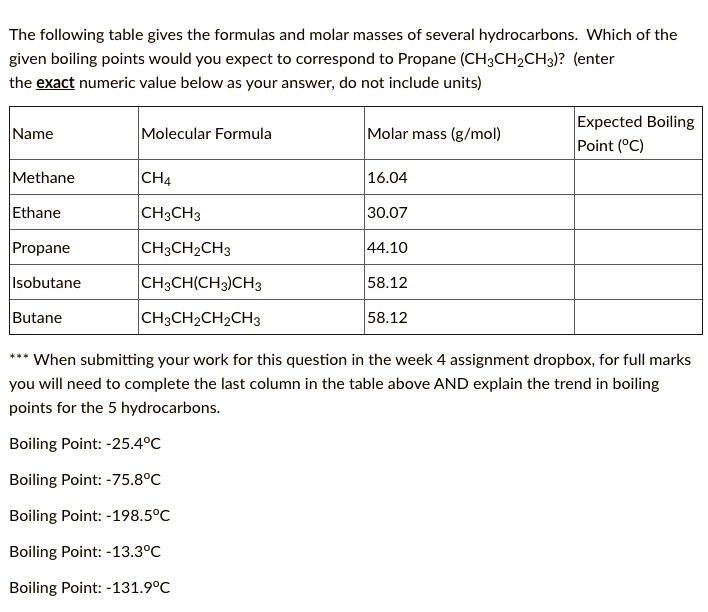
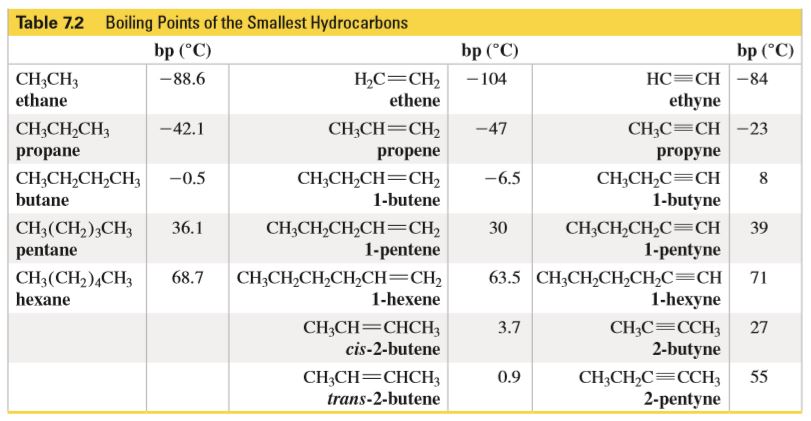
SOLVED: The following table gives the formulas and molar masses of several hydrocarbons: Which of the given boiling points would you expect to correspond to Propane (CH3CH2CH3)? (enter the exact numeric value

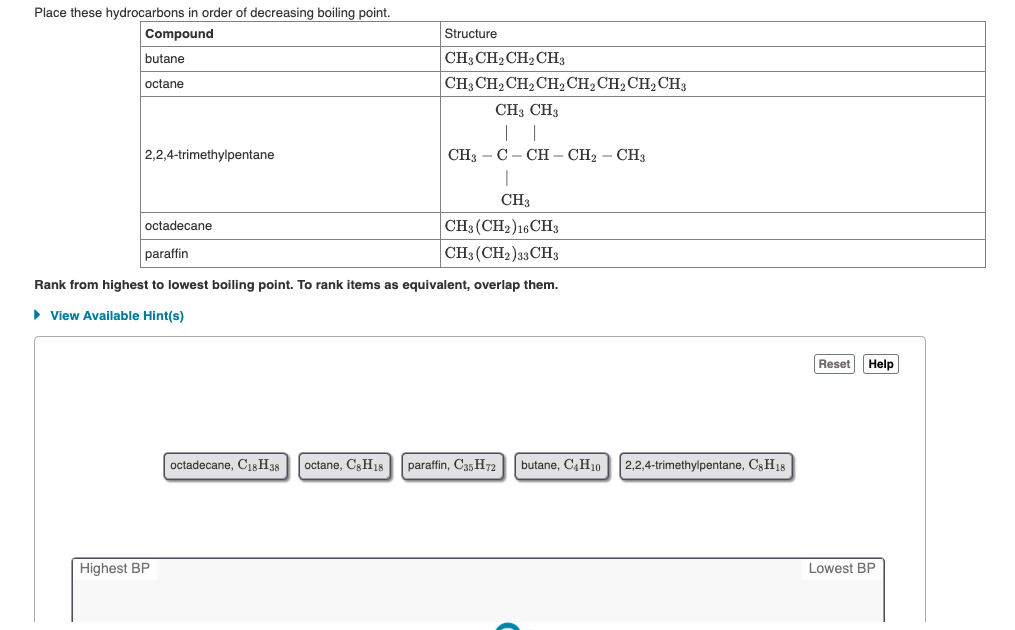
The increasing order of the boiling points for the following compounds is : (I)C2H5OH (II)C2H5Cl (III)C2H5CH3 (IV)C2H5OCH3
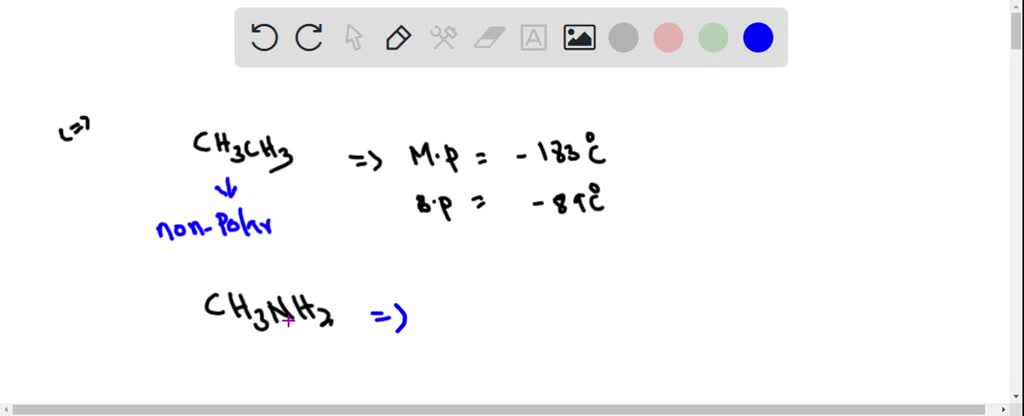
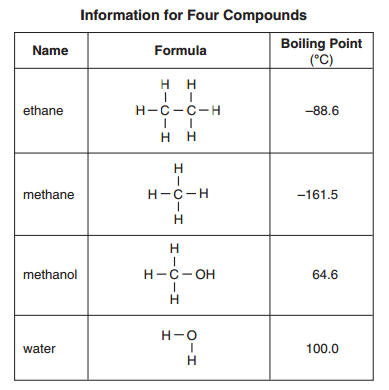
SOLVED: Ethane (CH3CH3) has a melting point of -183°Cand a boiling point of -89°C. Predict the melting point and boiling points for methylamine(CH3NH2)
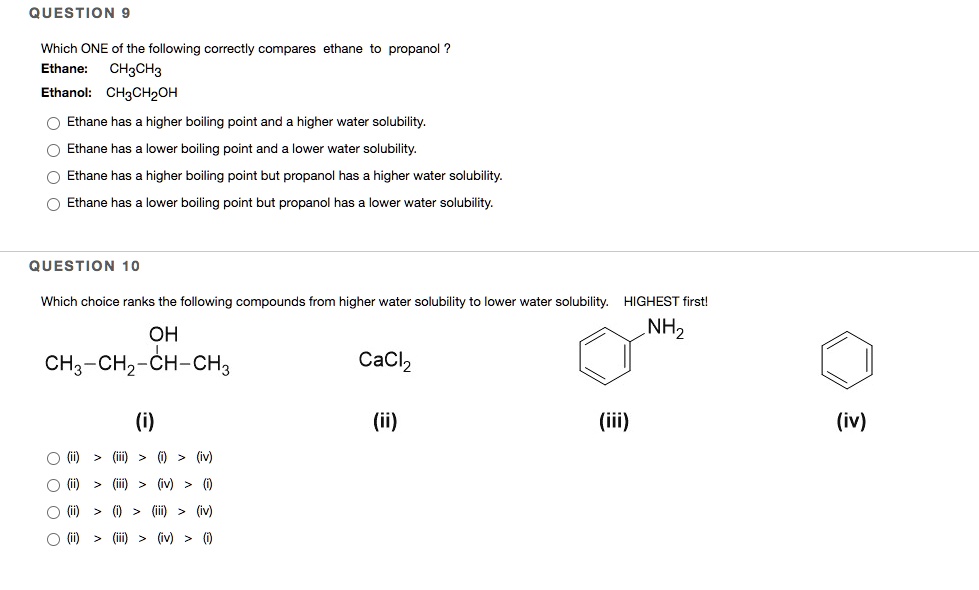
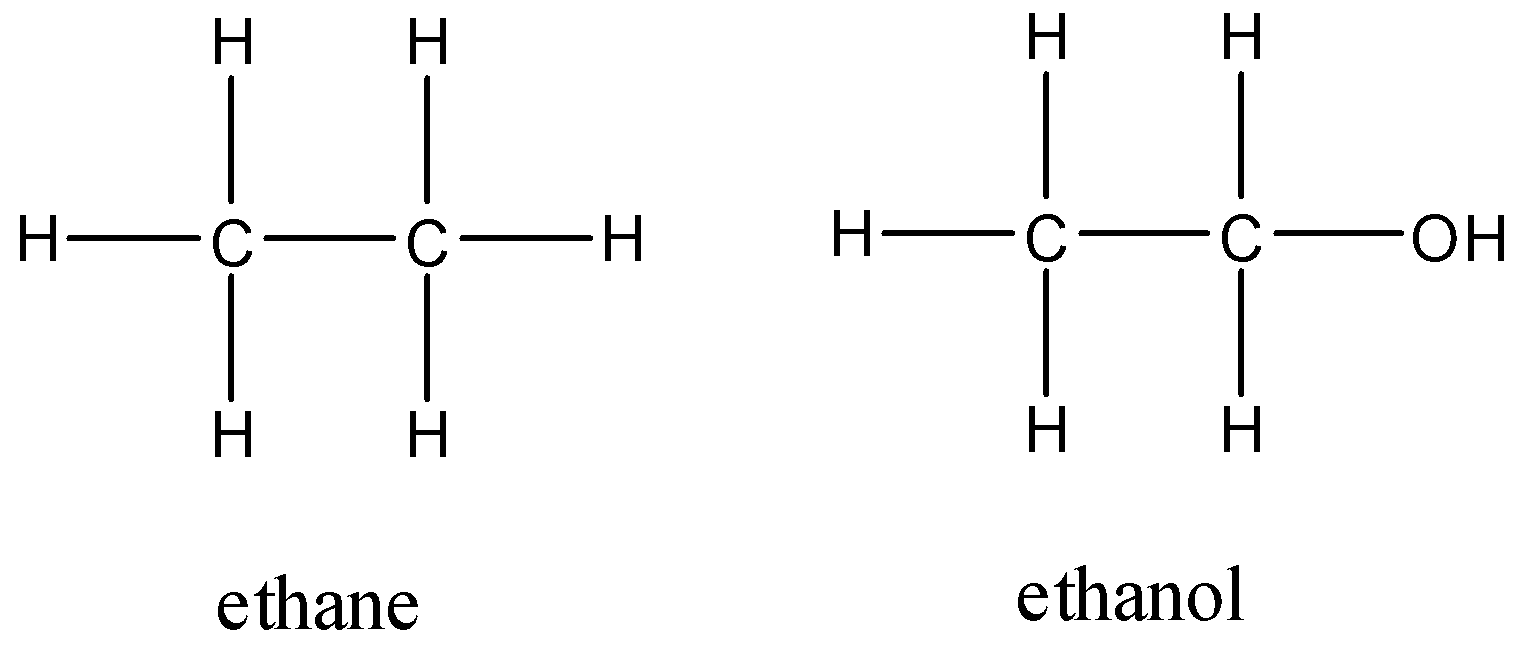
SOLVED: QUESTION Which ONE of the following correctly compares ethane Ethane: CH3CH3 Ethanol: CH:CHzOH propano Ethane has higher boiling point and higher water solubility: Ethane has lower boiling point and lower water